Recognizing and distinguishing between neutral and charged bonding patterns will be helpful in learning reaction mechanisms. During chemical reactions, it is common to have charge reactant, intermediates, and/or products. Organic molecules can also have positive or negative charges associated with them. In the beginning, it can be helpful to physically add the lone pair electrons.įor organic chemistry, the common bonding patterns of carbon, oxygen, and nitrogen have useful applications when evaluating chemical structures and reactivity. Since the lone pair electrons are often NOT shown in chemical structures, it is important to mentally add the lone pairs. Unshared electrons are also called ‘Lone Pairs’ and are shown as ‘:’ Double and triple bonds can also be communicated with lines as shown below.Ģ shared electrons form a single bond shown as ‘:’ or ‘–‘Ĥ shared electrons form a double bond shown as ‘::’ or ‘=’Ħ shared electrons form at triple bond shown as ‘:::’ or Lone pair (unshared) electrons are still shown as individual electrons. Line representations are only used for shared electrons. While it can be helpful initially to write the individual shared electrons, this approach quickly becomes awkward.Ī single line is used to represent one pair of shared electrons. Lewis structures, also known as Lewis-dot diagrams, show the bonding relationship between atoms of a molecule and the lone pairs of electrons in a molecule. Note: The review of general chemistry in sections 1.3 - 1.6 is integrated into the above Learning Objective for organic chemistry in sections 1.7 and 1.8. Now you score 5+15 POINTS by uploading your Pic and Downloading the Askiitians Toolbar respectively : Click here to download the toolbar.\)ĭraw, interpret, and convert between Lewis (Kekule), Condensed, and Bond-line Structures So help discuss any query on askiitians forum and become an Elite Expert League askiitian. Win exciting gifts by answering the questions on Discussion Forum. We are all IITians and here to help you in your IIT JEE preparation. 
Please feel free to post as many doubts on our discussion forum as you can. single bonded oxygen in NO 2 - FC = 6 - 6 - (2÷2) = -1.double bonded oxygen in NO 2 -: FC = 6 - 4 - (4÷2) = 0. 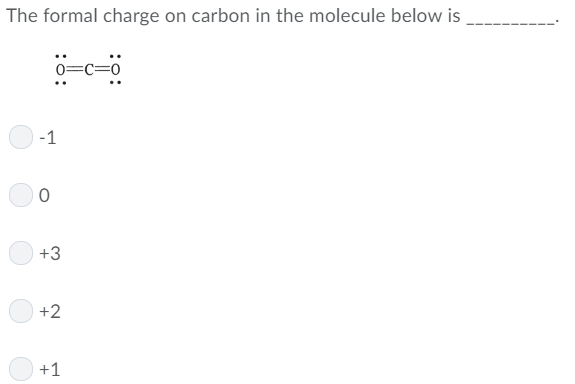
When determining the correct Lewis structure (or predominant resonance structure) for a molecule, the structure is chosen such that the formal charge (without sign) on each of the atoms is minimized. Where V is the number of valence electrons of the atom in isolation (atom in ground state) N is the number of non-bonding valence electrons on this atom in the molecule and B is the total number of electrons shared in covalent bonds with other atoms in the molecule. The formal charge of any atom in a molecule can be calculated by the following equation: Concepts of Physics by HC Verma for JEEĪ formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in a chemical bond are shared equally between atoms, regardless of relative electronegativity.IIT JEE Coaching For Foundation Classes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
